Calorimeter - Definition, Uses, Types, Application, Diagram, FAQs
Calorimetry is an important topic in physics that helps us understand how heat is transferred between different substances. On this page, you will learn the complete concept of calorimetry in a simple and clear way, starting from its definition, principle, and formula to how a calorimeter works in real life. You will also explore different types of calorimeters, their uses, and why calorimetry is widely applied in science, industry, and everyday situations. This page also includes step-by-step solved examples, important formulas, and easy explanations, making it useful for Class 11, Class 12, NEET, and JEE students.
This Story also Contains
- Definition of Calorimetry
- Principle of Calorimeter
- Uses of Calorimetry
- Types of Calorimeters
- Solved Examples Based on the Calorimetry Principle
- Summary

Definition of Calorimetry
Calorimetry is the process used to measure heat transfer during temperature change or phase change. A device called a calorimeter is used to calculate this heat exchange.
Principle of Calorimeter
The working of a calorimeter is based on the Law of Conservation of Energy.
Heat lost by the hotter body = Heat gained by the colder body
When a hot object is placed in contact with a cold object, heat flows from the hotter to the colder one until both reach the same temperature.
Calorimetry Formula
$$
Q=m c \Delta T
$$
Where:
- $\mathbf{Q}=$ Heat gained/lost
- $\mathbf{m}=$ Mass
- $\mathbf{c}=$ Specific heat capacity
- $\boldsymbol{\Delta} \mathbf{T}=$ Change in temperature
In calorimetry problems:
Heat lost $=$ Heat gained
Calorimeter - Explanation in Detail
The physical instrument or device which is used in the measurements of heat-related stuff (that is mainly used in the study of calorimetry). The construction of the calorimeter contains vessels made up of good conductors like metals. The composition of metallic vessels contains copper and aluminium in major. The contents of the metallic vessel are facilitated for stirring with the help of a stirrer. The heat loss in the stirrer is reduced with the help of an insulated jacket around the stirrer. The thermometer is allowed to be inserted through a small opening which is used to measure the thermal change that occurs inside the calorimeter.
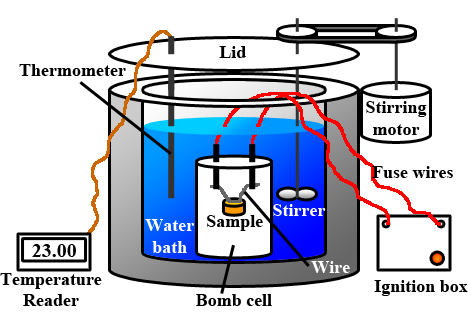
In the inner part of the calorimeter, some sample of fuel is used to burn. The metallic vessel contains water, when the fuel inside is burnt, the water gets heated. The heat loss of the fuel is converted into the heat gained by the water. That is, energy is conserved. To increase the accuracy of results, heat loss can be terminated by the insulation of the calorimeter instrument from the environmental conditions. The heat change of the water is measured by using the inserted thermometer. The readings are used to find the heat capacity of the water and the amount of energy conserved inside the fuel.
Uses of Calorimetry
Calorimetry is widely used in:
- Food industry to find calories in food
- Medical science to measure body metabolism
- Physics and chemistry labs to study reactions
- Engineering for designing cooling and heating systems
- Energy sector to test fuels
- Material science to study thermal properties
Types of Calorimeters
Different processes need different calorimeters.
Common types include:
1. Adiabatic Calorimeter - Prevents heat exchange with surroundings
2. Constant Pressure Calorimeter - Used in chemistry labs
3. Reaction Calorimeter - Measures heat during chemical reactions
4. Differential Scanning Calorimeter (DSC) - Used in material testing
5. Bomb Calorimeter - Used to find the calorific value of fuels
Recommended Topic Video
Solved Examples Based on the Calorimetry Principle
Example 1
The principle of calorimetry is based on:
1. Conservation of mass
2. Conservation of energy
3. Conservation of momentum
4. Both (1) and (2)
Solution: Heat lost $=$ Heat gained → law of conservation of energy.
Answer: (2)
Example 2 :An unknown metal (mass $\mathbf{1 9 2 ~ g}$ ) at $\mathbf{1 0 0}^{\boldsymbol{\circ}} \mathbf{C}$ is placed into a brass calorimeter (mass $\mathbf{1 2 8 ~ g}$ ) containing $\mathbf{2 4 0 ~ g}$ water at $\mathbf{8 . 4}^{\boldsymbol{\circ}} \mathbf{C}$. The final temperature is $\mathbf{2 1 . 5}^{\boldsymbol{\circ}} \mathbf{C}$. Find the specific heat s of the metal.
Options:
1. $458 \mathrm{~J} \mathrm{~kg}^{-1} \mathrm{~K}^{-1}$
2. $1232 \mathrm{~J} \mathrm{~kg}^{-1} \mathrm{~K}^{-1}$
3. $654 \mathrm{~J} \mathrm{~kg}^{-1} \mathrm{~K}^{-1}$
4. $916 \mathrm{~J} \mathrm{~kg}^{-1} \mathrm{~K}^{-1}$
(Specific heat: brass $=394 \mathrm{~J} \mathrm{~kg}^{-1} \mathrm{~K}^{-1}$, water $=4200 \mathrm{~J} \mathrm{~kg}^{-1} \mathrm{~K}^{-1}$ )
Solution :
Convert masses to kg: m_m = 0.192 kg , m_brass = 0.128 kg , m_water = 0.240 kg .
Heat lost by metal = Heat gained by brass + water:
$
0.192 s(100-21.5)=0.128 \times 394(21.5-8.4)+0.240 \times 4200(21.5-8.4)
$
Solving gives $s \approx 916 \mathrm{~J} \mathrm{~kg}^{-1} \mathrm{~K}^{-1}$.
Answer: (4)
Example 3
A liquid of mass $m$ and specific heat $C$ is at temperature $2 T$. Another liquid of mass $m / 2$ and specific heat $\mathbf{2 C}$ is at temperature T. They are mixed. The final temperature is:
Options:
1. $\frac{2 T}{3}$
2. $\frac{8 T}{5}$
3. $\frac{3 T}{5}$
4. $\frac{3 T}{2}$
Solution: Use $\theta_{\text {mix }}=\frac{m_1 c_1 \theta_1+m_2 c_2 \theta_2}{m_1 c_1+m_2 c_2}$.
$
\theta=\frac{m C \cdot 2 T+\frac{m}{2} \cdot 2 C \cdot T}{m C+\frac{m}{2} \cdot 2 C}=\frac{2 m C T+m C T}{2 m C}=\frac{3 T}{2} .
$
Answer: (4)
Example 4
Ice at $\mathbf{- 2 0} \mathbf{~ C}^{\boldsymbol{\circ}} \mathbf{C}$ is added to $\mathbf{5 0 ~ g}$ water at $\mathbf{4 0}^{\boldsymbol{\circ}} \mathbf{C}$. After reaching $\mathbf{0}^{\boldsymbol{\circ}} \mathbf{C}$, $\mathbf{2 0 ~ g}$ ice remains unmelted. The amount of ice added was:
Options:
1. 100 g
2. 50 g
3. 40 g
4. 60 g
(Specific heats: water $4.2 \mathrm{~J} / \mathrm{g}^{\circ} \mathrm{C}$, ice $2.1 \mathrm{~J} / \mathrm{g}^{\circ} \mathrm{C}$; latent heat of fusion $=334 \mathrm{~J} / \mathrm{g}$ )
Solution: Let total ice $=\mathrm{mg}$. Heat taken by ice to reach $0^{\circ} \mathrm{C}$ and melt (only $\mathrm{m}-20$ melts) equals heat lost by water cooling $40 \rightarrow 0^{\circ} \mathrm{C}$.
Equation:
$
20 \times 2.1 \times m+(m-20) \times 334=50 \times 4.2 \times 40
$
Solving yields $m \approx 40 \mathrm{~g}$.
Answer: (3)
Example 5 : 5 g water at $10^{\circ} \mathrm{C}$ is mixed with 5 g water at $40^{\circ} \mathrm{C}$. Final temperature is:
Options:
1. $20^{\circ} \mathrm{C}$
2. $25^{\circ} \mathrm{C}$
3. $30^{\circ} \mathrm{C}$
4. $35^{\circ} \mathrm{C}$
Solution: Equal masses and same specific heat → average temperature:
$
\theta=\frac{10+40}{2}=25^{\circ} \mathrm{C} .
$
Answer: (2)
Summary
Calorimetry is the study of heat transfer during physical changes or chemical reactions, governed by the principle of conservation of energy. A calorimeter measures heat exchange between substances, such as the heat lost by a hotter object being equal to the heat gained by a cooler one. This principle has applications in various fields, from everyday activities like boiling water to complex scientific and industrial processes.
Frequently Asked Questions (FAQs)
There are several types of calorimeters based on their design and use. The main types are:
Adiabatic calorimeter
Constant pressure calorimeter
Bomb calorimeter
Reaction calorimeter
Differential scanning calorimeter (DSC)
Using a calorimeter involves simple steps:
Take the calorimeter and add a known mass of water.
Measure and record the initial temperature of the water.
Heat the solid or liquid sample to a known temperature.
Quickly transfer the hot sample into the calorimeter containing water.
Close the lid to prevent heat loss and stir gently.
Note the final temperature once the system reaches thermal equilibrium.
Use the formula Heat lost = Heat gained to calculate specific heat, heat change, or energy value.
A calorimeter is a device used to measure the amount of heat absorbed or released during physical or chemical processes. It works on the principle of conservation of energy—heat lost by a hot substance is equal to the heat gained by a cold substance.
The concept of calorimetry is used in the measurement of the change in thermal condition of anybody. The study of the measurement of the changes that occur in the state variable of anybody to find the amount of heat transfer that happens along with state transfer is defined as calorimetry. A calorimeter is used to measure and study the calorimetry.
The construction of the calorimeter contains vessels made up of good conductors like metals. The composition of metallic vessels contains copper and aluminium in major. The contents of the metallic vessel are facilitated for stirring with the help of a stirrer. The heat loss in the stirrer is reduced with the help of an insulated jacket around the stirrer. The thermometer is allowed to be inserted through a small opening which is used to measure the thermal change that occurs inside the calorimeter. Inside the calorimeter, some sample of fuel is used to burn. The metallic vessel contains water, when the fuel inside is burnt, the water gets heated. The heat loss of the fuel is converted into the heat gained by the water. That is, energy is conserved. To increase the accuracy of results, heat loss can be terminated by the insulation of the calorimeter instrument from the environmental conditions. The heat change of the water is measured by using the inserted thermometer. The readings are used to find the heat capacity of the water and the amount of energy conserved inside the fuel.
The different types of calorimeters are
Adiabatic calorimeters
Constant pressure calorimeter
Reaction calorimeters
Differential scanning calorimeter
Bomb calorimeters
The main principle of the calorimeter is the law of conservation of energy. the heat transformations occur from the higher temperature body to the lower temperature body. This transformation occurs until both bodies attain thermal equilibrium. The heat is released by the higher temperature body and the heat is absorbed by the lower-temperature body.
Let us discuss the application of calorimeter or The real-life application of calorimetry can be seen in different fields of industries and laboratories. Some examples of the practical calorimeters and their uses are listed below:
Differential scanning calorimeters are used to find the change in any product's formula and its effects
A reaction calorimeter is used to measure the heat generated by the sensors in reactors.
A constant pressure calorimeter helps us to measure the enthalpy change in both physical and chemical methods.
The adiabatic calorimeter is used to evaluate the runtime reactions.
Bomb calorimeters are widely used in different kinds of fields. Some of the fields are analysis of waste products, manufacture of cement, analysis of fuel like coal and explosives, food and nutrition, research in animal feed and so on. For example, the Oxygen bomb calorimeter has its usage in the field of food testing by calculating the amount of heat in food which further helps to determine the calorie of the food.

