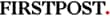the increasing order ofacidity of H2o2, H2O,co2
New: JEE Main 2025 Session 1 Result OUT; Check Now | Rank Predictor
JEE Main 2025: College Predictor | Marks vs Rank vs Percentile | Top NITs Cutoff
JEE Main 2025: January Session Official Question Paper for All Shifts | Exam Analysis
JEE Main 2025: High Scoring Topics | Sample Papers | Mock Tests | PYQs
The increasing order of acidity is
H2O < H2O2 < CO2
The reason is
CO2is a stronger acid thanH2O2since it liberatesH2O2fromBaO2. Water, of course, is a weaker acid thanH2O2.
Hope it helps
The correct increasing order stands as H2O<H2O2<CO2.
I hope this helps
Related Questions
Know More about
Joint Entrance Examination (Main)
Eligibility | Application | Preparation Tips | Question Paper | Admit Card | Answer Key | Result | Accepting Colleges
Get Updates Brochure
Your Joint Entrance Examination (Main) brochure has been successfully mailed to your registered email id “”.